Davidson Lab
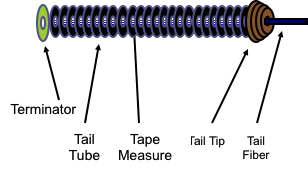
Phage Tails
Ninety five percent of phages on earth utilize a structure known as a tail to bind to the surface of bacteria for the initiation of infection. Tails impart host specificity to phages by binding particular proteins or sugars on the cell surface, which are the most variable components of bacteria. They also mediate the injection of the phage genome through the bacterial membranes and cell wall into the cell interior. Phage tails are tremendously sophisticated nanomachines with the ability to deliver genomes of more than half a million base pairs. The power of phage tails is proven by their ubiquity on earth and by the adaptation of phage tails into machineries utilized by bacteria for toxin transfer into both bacteria and eukaryotic cells, as exemplified by Photorhabdus Virulence Cassettes and Type VI secretion systems. These are both tail-derived entities that can kill eukaryotic cells.
In addition to investigating the structure and function of phage tails, we also research naturally occurring bacterial-killing nanomachines, called tailocins, which have evolved from phage tails. We aim to develop tailocins as agents for microbiome manipulation and as alternatives to antibiotics. Tailocins have great advantages for this purpose because their killing specificity can be precisely engineered against any bacterium, even ones that have not been cultured. In addition, they can be produced in large quantities from expression plasmids in amenable lab strains, such as E. coli.